
By Carolynn Grimes
ThePlasticSurgeryChannel.com
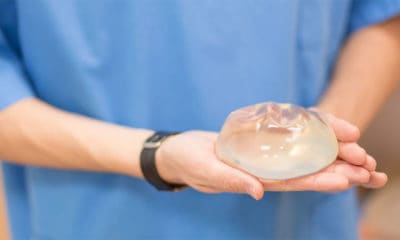
There’s a new competitor in the silicone breast implant industry that will lead to more choices for women. The Food and Drug Administration has granted conditional approval of a new silicone gel breast implant. California based, Sientra, announced it will offer the implants in a full range of different shapes and sizes. Until now only two companies, Mentor and Allergan, have manufactured and sold silicone implants in the U.S.
Last year, according to the American Society of Plastic Surgeons (ASPS), plastic surgeons performed more than 300,000 breast augmentation procedures in the U.S., making it the most popular cosmetic surgery for women. The surgery consists of surgically placing an implant under the breast tissue or chest muscle to reconstruct or augment the breast.
So what does this new competition mean to consumers?
Dr. Brett Snyder, a board certified plastic surgeon from Jacksonville, Florida says, “Sientra’s entry into the gel implant market means more competition, more products and potentially lower prices.” Chief Executive Officer of Sientra, Hani Zeini, states “Sientra has successfully broken the existing duopoly in the U.S. by offering surgeons and patients a new choice.’’
Board certified plastic surgeon, Dr. Peter Fodor from Beverly Hills, California says, “All implants have their respective benefits and shortcomings.” He says, having more choices helps patients in their implant selection process. “Of course, this is contingent on patients being properly informed about what is available to them,” according to Fodor.
Dr. Snyder agrees and says, “The decision on which implant to use will still be based on the doctors evaluation of the patient.” He says, while some implants made by different companies are similar, some of Sientra’s implants have different characteristics than those currently available. “Based on the surgeon’s evaluation of the patient, he/she may recommend one implant over another due to these different characteristics,” says Snyder.
Patients aren’t the only ones who will benefit from more choices.

Sientra’s CEO tells The Plastic Surgery Channel, “approval is a net gain for the patients and physician as it provides previously unavailable choices to best address the individual needs of the patients while providing expanded choice pallet for plastic surgeons in addressing the best options for their patients…a true win-win for everyone!”
Surgeons with ThePlasticSurgeryChannel.com remind woman seeking breast augmentation, to consult with a board certified plastic surgeon to determine the best implant for them.












Facebook
Twitter
Instagram
YouTube
RSS